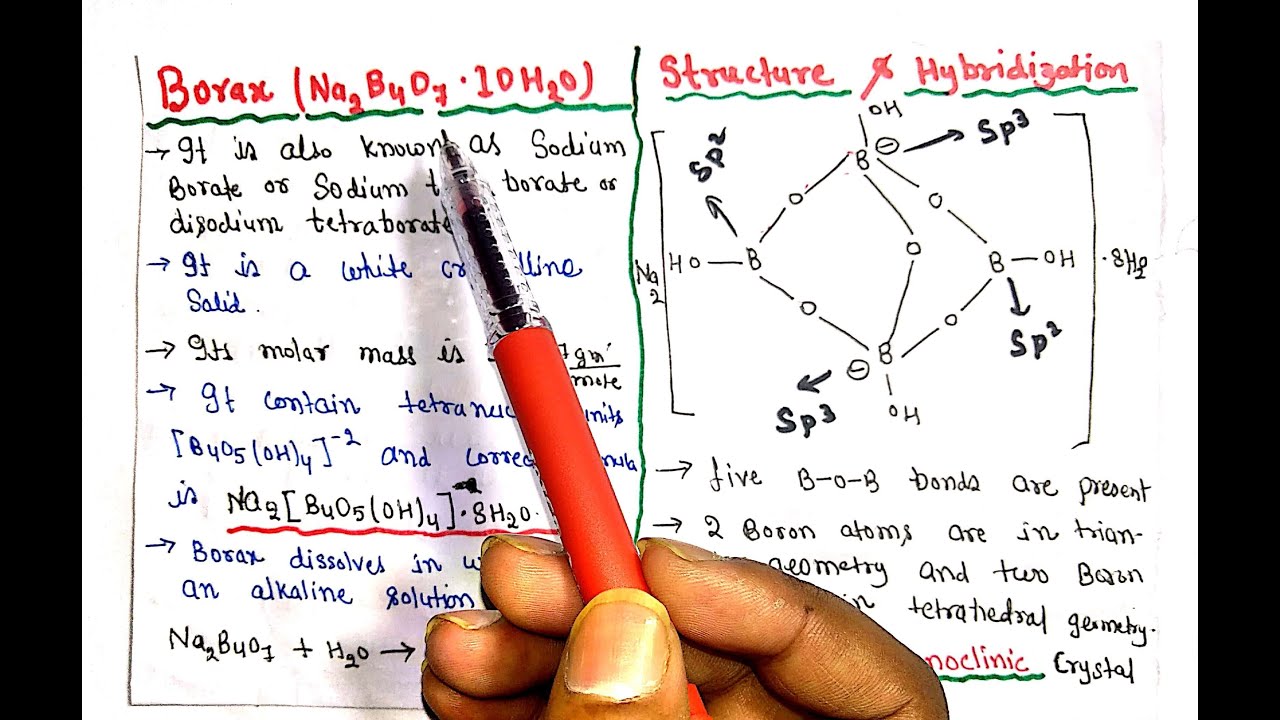
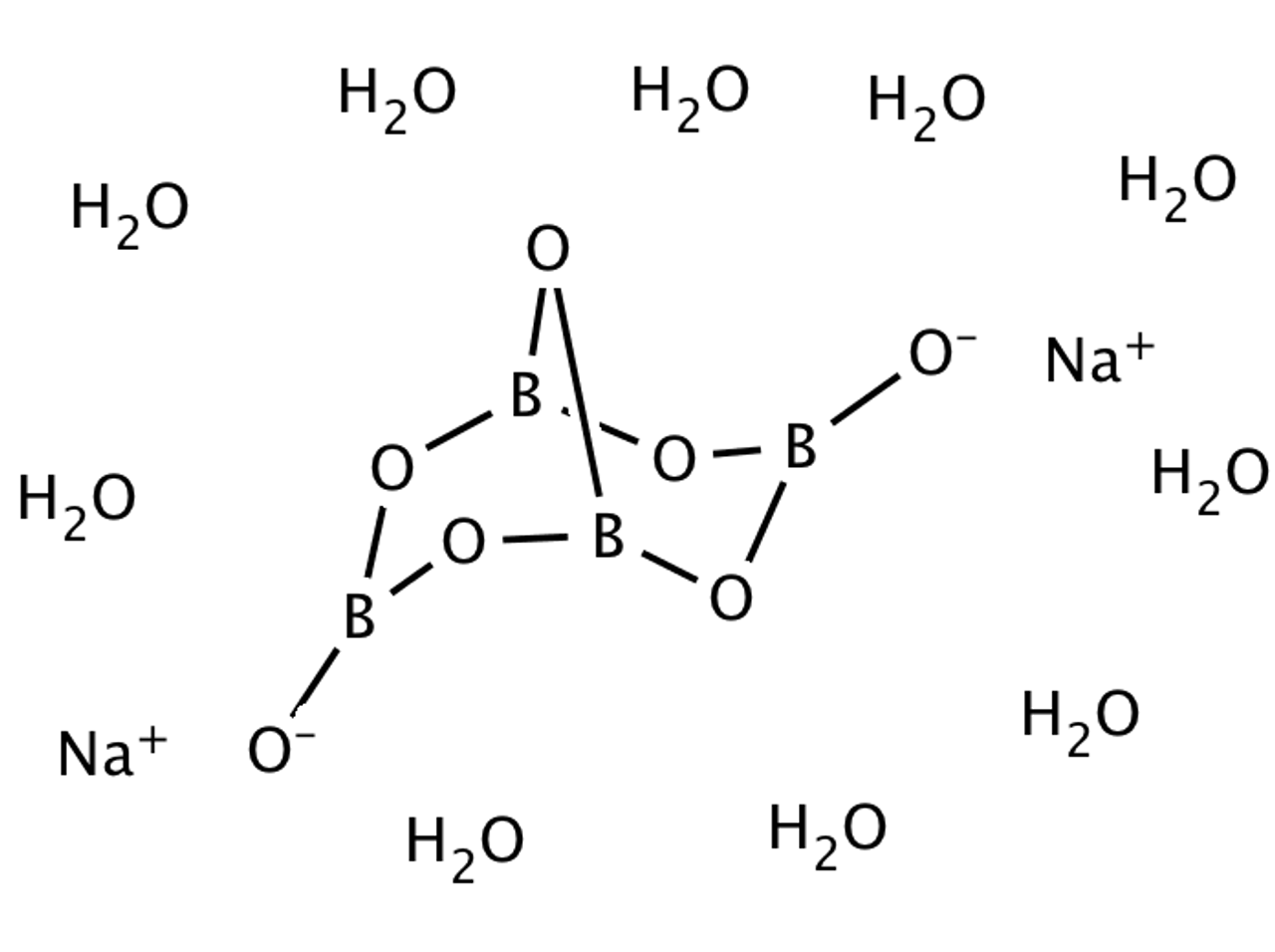
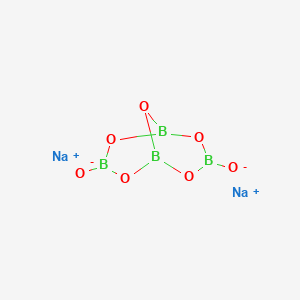
Borax has the formula Na2B4O7. 10H2O. It is a strong base in aqueous solution because OH^- ions are produced by reaction with water. (B4O7^2 - + 7H2O → 4H3BO3 + 2OH^-). How
Calculate the % of boron (B) in borax Na2 B4 O7 . 10H2O. (H = 1, B = 11, O = 16, Na = 23) - Sarthaks eConnect | Largest Online Education Community

BFC BORAX LR - 5kg, DI-SODIUM TETRABORATE (Na2B4O7.10H2O) CAS No.1303-96-4 : Amazon.in: Industrial & Scientific