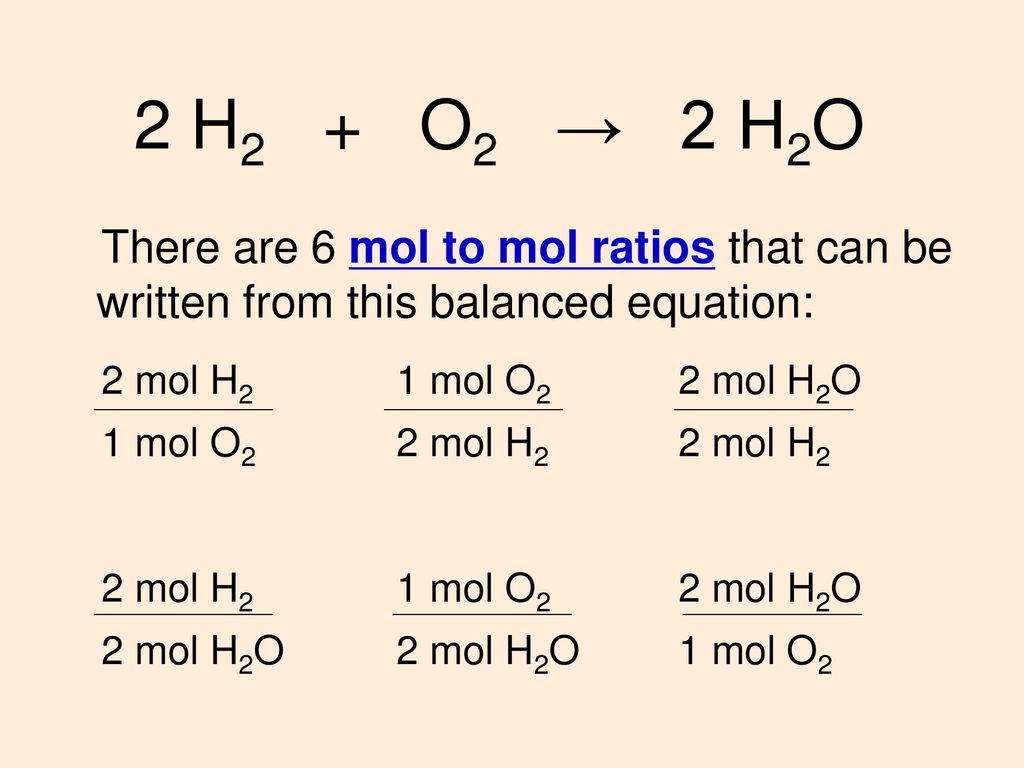
SOLVED: How many moles of hydrogen atoms are present in a sample that contains 3.64 moles of water, H2O? answer=mol H How many moles of H2O molecules are present in a sample

The molar mass of H2O is 18.01 g/mol . The molar mass of 02 is 32.00 g/mol What is the mass of H20 in - Brainly.com

Wet air containing 4.0 mole% water vapor is passed through a column of calcium chloride pellets. The pellets absorb 97.0% of the water and none of the other constituents of the air.

Question Video: Calculating the Mass of Water Produced Given the Masses of Oxygen and Hydrogen | Nagwa
![Calculate the depression in the freezing point of water when 10 g of CH3CH2CHClCOOH is added to 250 g of water. [ Ka = 1.4 × 10^-3, Kf = 1.86 K kg mol^-1 ] Calculate the depression in the freezing point of water when 10 g of CH3CH2CHClCOOH is added to 250 g of water. [ Ka = 1.4 × 10^-3, Kf = 1.86 K kg mol^-1 ]](https://d1hhj0t1vdqi7c.cloudfront.net/v1/ekdmSWJoaW9GWjA=/sd/)
Calculate the depression in the freezing point of water when 10 g of CH3CH2CHClCOOH is added to 250 g of water. [ Ka = 1.4 × 10^-3, Kf = 1.86 K kg mol^-1 ]